|
|
Post by kammy on Jul 19, 2011 20:19:50 GMT -5
|
|
|
|
Post by kammy on Jul 22, 2011 21:00:52 GMT -5
Massive ice island drifts toward Canada www.msnbc.msn.com/id/43848004/ns/us_news-environment/7/21/2011 "A Manhattan-sized chunk of ice that broke off a glacier in Greenland nearly a year ago is drifting toward the coast of Newfoundland, Canada — providing a stunning sight to scientists and curiosity-seekers but also posing a potential threat to ships." |
|
|
|
Post by skyship on Aug 15, 2011 22:46:45 GMT -5
|
|
|
|
Post by kammy on Aug 18, 2011 17:24:37 GMT -5
|
|
|
|
Post by kammy on Aug 20, 2011 7:52:35 GMT -5
"A wide variety of solutions can give rise to them; for example, coacervates form spontaneously when a protein, such as gelatin, reacts with gum arabic." Brings us back to a protein. I said somewhere back based on somebody's research about a missing protein - that I thought ferrodoxin was the missing protein. So, is our main culprit a protein or overload of that protein that causes everything to be set into action to cause evident disease or the disease multiplying? Or, a gelatin (polysaccharide sugar) (does it contain protein? ferrodoxin?) is causing the shell walls of the spheres to be formed - creating the spheres/coacervates within us? Questions: What's the proteins involved in gelatins? Does polysaccharide sugar contain proteins and/or is it possible that ferrodoxin is now being introduced via the atmosphere/rain/soil into the sugar? What's the relationship between carbon and ferrodoxin? How or what would cause the coacervates to be becoming different from each other, capable of producing different pathogens? ""A wide variety of solutions can give rise to them;..." When you look at ferrodoxin - it takes you to: en.wikipedia.org/wiki/Electron_transfer"Numerous biological processes involve ET reactions, including oxygen binding, photosynthesis, respiration, and detoxification routes. Additionally, the process of energy transfer can be formalized as a two electron exchange (two concurrent ET events in opposite directions) in case of small distances between the transferring molecules. ET reactions commonly involve transition metal complexes," Which takes us to inner and outer sphere electron transfer... redox - en.wikipedia.org/wiki/Inner-sphere_electron_transfer |
|
|
|
Post by kammy on Aug 20, 2011 7:53:20 GMT -5
Q> "How or what would cause the coacervates to be becoming different from each other, capable of producing different pathogens?"
"Coarcervates are interesting in that not only do they provide a locally segregated environment but also that their boundaries allow the selective adsorption of simple organic molecules from the surrounding medium."
So... thinking out loud. You know the biopesticides that allow a powder to be turned into a beneficial nematode. Just add water and presto - thousands of nematodes appear - it's magic. Wonder how they work, exactly? I know we looked into it a little bit on the LB 'Baculovirus' thead... probably didn't dig deep enough.
What I'm thinking is that these various pathogens that we've come into contact with might be floating around in our blood streams in the form of... minute particles?, atoms?, nanotechnology creations?, of course, the viruses from the vaccines and all the other trash... are not in a cellular form such as our spheres.
And then some sort of combination of metals, proteins, (maybe enzymes?), definitely some sort of electron transfer cause the Coarcervates to be formed - but, somehow - they are selective in what they choose to take in. Because we see that all spheres appear equal and alike in the beginning but then one may burst into a white 'fungus', another into a black 'fungus', another into a red and blue hyphae/fibered 'fungus' or bacteria?... synthetic mix?. Another may be a holder for a baculoviral tardigrade alone or a calcite crystal formation that resembles HIV6. Another goes through the motions of how a baculoviral system is known to operate, expelling carbon-coated occlusion bodies that resemble stacked calcite crystals, and other crystal shapes, that can hold a synthetic baculoviral insect or parasite. Another may just produce the biofilm. The biofilm then becomes like a protective womb that can hold any of the above. And, they all reproduce using similar methods within their groups but each is using a different method.
That's what we're dealing with... a complicated, inter-woven synthetic system that is hard to detect because modern-day testing equipment on the basic levels (doctors/hospitals/labs) is not up with the technology and for the average person - if they are able to figure out how to 'see' it, it's almost impossible to define. Plus, a lot of the key information is hidden or been withdrawn.
|
|
|
|
Post by kammy on Aug 20, 2011 7:54:27 GMT -5
"J. D. Bernal commented that they are "the nearest we can come to cells without introducing any biological – or, at any rate, any living biological – substance." However, the lack of any mechanism by which coacervates can reproduce leaves them far short of being actual living systems."
So, if our sphere is not a coacervates then it is a 'souped-up' cousin because we're seeing 'biologicals' inside them and they can reproduce in many prolific ways. Maybe, looking to see who did what and what was done in research with coacervates might lend us some clues?
|
|
|
|
Post by kammy on Aug 20, 2011 7:55:40 GMT -5
Q. What's the relationship between carbon and ferrodoxin? Search - carbonaceous ferrodoxin www.ncbi.nlm.nih.gov/pmc/articles/PMC2025642/"The current energy usage in the United States is heavily dependent (66%) on carbonaceous fossil fuel sources (coal, heating oil, natural gas), with minority contributions from nuclear, hydrological, or alternatives. Several species of green algae have been shown to possess this ability (7,8), including Scenedesmus obliquus (9), Chlorella fusca (10), and the free-living unicellular alga Chlamydomonas reinhardtii (11). Hydrogen production in the latter has been particularly well studied (12–18), and the requirements for maturation and heterologous expression of its two [FeFe] hydrogenases identified (19–21)." ======== Ferredoxins in the evolution of photosynthetic systems from anaerobic bacteria to higher plants www.springerlink.com/content/lh11016281840665/ |
|
|
|
Post by kammy on Aug 20, 2011 7:56:08 GMT -5
bacterial ferredoxin? New perspectives on bacterial ferredoxin evolution www.springerlink.com/content/hj16t773161h5vx4/"A number of ferredoxin sequences do not fit into any of the groups described above. Based on sequence properties, these sequences can be separated into three groups: a group containing Methanosarcina barkeri ferredoxin and Desulfovibrio desulfuricans ferredoxin II, a group containing Desulfovibrio gigas ferredoxin and Clostridium thermoaceticum ferredoxin, and a group containing Desulfovibrio africanus ferredoxin I and Bacillus stearothermophilus ferredoxin. The last two groups differ from all of the other bacterial ferredoxins in that they bind only one FeS cluster per polypeptide, whereas the others bind two. Sequence examination indicates that the second binding site has been either partially or completely lost from these ferredoxins. Methanosarcina barkeri ferredoxin and Desulfovibrio desulfuricans ferredoxin II are of interest because, of all the ferredoxins whose sequences are presently known, they show the strongest evidence of internal gene duplication. " |
|
|
|
Post by kammy on Aug 20, 2011 7:57:25 GMT -5
I'm going to call them gas water carbon molecules for now or gas hydrocarbons or hydrocarbons, for short.
|
|
|
|
Post by skyship on Aug 20, 2011 13:58:25 GMT -5
The Pahs. Right direction, here is a list, so we know what we are dealing with. Polycyclic aromatic hydrocarbons (PAHs), also known as poly-aromatic hydrocarbons or polynuclear aromatic hydrocarbons, are potent atmospheric pollutants that consist of fused aromatic rings and do not contain heteroatoms or carry substituents.[2] Naphthalene is the simplest example of a PAH. en.wikipedia.org/wiki/Polycyclic_aromatic_hydrocarbon#List_of_PAHsFound in outer space too, however, of different constituency. RELEASE : 10-65AR NASA Reveals Key to Unlock Mysterious Red Glow in Space Messier-82 galaxy Above image combines visible-infrared Spitzer Space Telescope images of the galaxy Messier-82. The red streaming away from the galaxy into intergalactic space traces the infrared emission from PAHs. Click image for full-resolution. interstellar nebula Above image is an interstellar nebula, showing the emission from PAHs in red, some PAH molecular structures and the interstellar PAH infrared signature. Click image for full-resolution. MOFFETT FIELD, Calif. -- NASA scientists created a unique collection of polycyclic aromatic hydrocarbon (PAH) spectra to interpret mysterious emission from space. Because PAHs are a major product of combustion, remain in the environment, and are carcinogenic, the value of this PAH spectral collection extends far beyond NASA and astronomical applications. For years, scientists have been studying a mysterious infrared glow from the Milky Way and other galaxies, radiating from dusty regions in deep space. By duplicating the harsh conditions of space in their laboratories and computers, scientists have identified the mystifying infrared emiters as PAHs. PAHs are flat, chicken-wire shaped, nano-sized molecules that are very common on Earth. “PAHs in space are probably produced by carbon-rich, giant stars. A similar process produces soots here on Earth,” said Louis Allamandola, an astrochemistry researcher at NASA’s Ames Research Center, Moffett Field, Calif. “Besides astronomical applications, this PAH database and software can be useful as a new research tool for scientists, educators, policy makers, and consultants working in the fields of medicine, health, chemistry, fuel composition, engine design, environmental assessment, environmental monitoring, and environmental protection.” www.nasa.gov/centers/ames/news/releases/2010/10-65AR.html |
|
|
|
Post by kammy on Aug 23, 2011 12:22:36 GMT -5
Yes, we agree. I'm sure it's some sort of modified PAH but for now - this will do... AND... a lot has been written about them for reference.
We're looking for a fiber/hyphae? to PAH or PAH to fiber/hyphae or combination possible of happening. The others I have separated and am just calling them 'particulate matter' for now.
|
|
|
|
Post by kammy on Aug 23, 2011 12:28:16 GMT -5
Polycyclic Aromatic Hydrocarbons (PAHs) Clinical Assessment www.atsdr.cdc.gov/csem/csem.asp?csem=13&po=12Signs and Symptoms—Acute Exposure "Acute effects attributed to PAH exposure, such as headache, nausea, respiratory and dermal irritation, are probably caused by other agents. Since PAHs have low acute toxicity, other more acutely toxic agents probably cause the acute symptoms attributed to PAHs. Hydrogen sulfide in roofing tars and sulfur dioxide in foundries are examples of concomitant, acutely toxic contaminants. Naphthalene, the most abundant constituent of coal tar, is a skin irritant, and its vapors may cause headache, nausea, vomiting, and diaphoresis" Signs and Symptoms—Chronic Exposure "Effects reported from occupational exposure to PAHs include chronic bronchitis, chronic cough irritation, bronchogenic cancer, dermatitis, cutaneous photosensitization, and pilosebaceous reactions. Reported health effects associated with chronic exposure to coal tar and its by-products (e.g., PAHs). Skin: erythema, burns, and warts on sun-exposed areas with progression to cancer. The toxic effects of coal tar are enhanced by exposure to ultraviolet light. Eyes: irritation and photosensitivity. Respiratory system: cough, bronchitis, and bronchogenic cancer. Gastrointestinal system: leukoplakia, buccal-pharyngeal cancer, and cancer of the lip. Hematopoietic system: leukemia (inconclusive) and lymphoma. Genitourinary system: hematuria and kidney and bladder cancers. Although researchers have examined PAHs directly in the blood and tissues of experimental animals, these methods have not been widely used for human samples. The high costs of testing and limited knowledge of the significance of background levels in humans limit the clinical usefulness of such tests."  |
|
|
|
Post by kammy on Aug 23, 2011 12:40:11 GMT -5
What Are the Routes of Exposure for PAHs? www.atsdr.cdc.gov/csem/csem.asp?csem=13&po=6excerpt "PAH exposure through air, water, soil, and food sources occurs on a regular basis for most people. Routes of exposure include ingestion, inhalation, and dermal contact in both occupational and non-occupational settings. Some exposures may involve more than one route simultaneously, affecting the total absorbed dose (such as dermal and inhalation exposures from contaminated air). All non-workplace sources of exposure such as diet, smoking, and burning of coal and wood should be taken into consideration. Cigarette smoking and environmental tobacco smoke are other sources of air exposure. Smoking one cigarette can yield an intake of 20–40 ng of benzo (a) pyrene [Phillips 1996; O’Neill et al. 1997]. Smoking one pack of unfiltered cigarettes per day yields 0.7 µg/day benzo(a)pyrene exposure. Smoking a pack of filtered cigarettes per day yields 0.4 µg/day [Sullivan and Krieger 2001]. Environmental tobacco smoke contains a variety of PAHs, such as benzo(a)pyrene, and more than 40 known or suspected human carcinogens. Side-stream smoke (smoke emitted from a burning cigarette between puffs) contains PAHs and other cytotoxic substances in quantities much higher than those found in mainstream smoke (exhaled smoke of smoker) [Jinot and Bayard 1996; Nelson 2001]. Water PAHs can leach from soil into water. Water contamination also occurs from industrial effluents and accidental spills during oil shipment at sea. Concentrations of benzo(a)pyrene in drinking water are generally lower than those in untreated water and about 100-fold lower than the U.S. Environmental Protection Agency’s (EPA) drinking water standard. (EPA’s maximum contaminant level [MCL] for benzo(a)pyrene in drinking water is 0.2 parts per billion [ppb].) Soil Soil contains measurable amounts of PAHs, primarily from airborne fallout. Documented levels of PAHs in soil near oil refineries have been as high as 200,000 micrograms per kilogram (µg/kg) of dried soil. Foodstuffs In non-occupational settings, up to 70% of PAH exposure for a non-smoking person can be associated with diet [Skupinska et al. 2004]. PAH concentrations in foodstuffs vary. Charring meat or barbecuing food over a charcoal, wood, or other type of fire greatly increases the concentration of PAHs. For example, the PAH level for charring meat can be as high as 10–20 µg/kg [Phillips 1999]. Charbroiled and smoked meats and fish contain more PAHs than do uncooked products, with up to 2.0 µg/kg of benzo(a)pyrene detected in smoked fish. Tea, roasted peanuts, coffee, refined vegetable oil, cereals, spinach, and many other foodstuffs contain PAHs. Some crops, such as wheat, rye, and lentils, may synthesize PAHs or absorb them via water, air, or soil. Other Sources of Exposure PAHs are found in prescription and nonprescription coal tar products used to treat dermatologic disorders such as psoriasis and dandruff [Van Schooten 1996]. PAHs and their metabolites are excreted in breast milk, and they readily cross the placenta. Anthracene laxative use has been associated with melanosis of the colon and rectum. Background Exposures Pyrene is commonly found in PAH mixtures, and its urinary metabolite, 1-hydroxypyrene, has been used as an indicator of exposure to PAH chemicals. Higher levels have been noted for residents of industrialized urban areas than in rural or suburban settings [Adonis et al. 2003; Kanoh et al. 1993; Kuo et al. 2004]. Many-fold higher levels can be found in workers from certain occupations [Jacob and Seidel 2002], including aluminum smelting [Alexandrie et al. 2000]; diesel engine mechanics [Adonis et al. 2003; Kuusimaki et al. 2004]; taxi, bus, and truck drivers [Chuang et al. 2003; Hansen et al. 2004; Kuusimaki et al. 2004]; painters [Lee et al. 2003], boilermakers [Mukherjee et al. 2004]; toll booth operators [Tsai et al. 2004]; traffic police [Merlo et al. 1998] and coke oven plant workers [Lu et al. 2002; Serdar et al. 2003; Siwinska et al. 2004]. Tobacco smoking leads to higher levels in smokers [Chuang et al. 2003; Adonis et al. 2003; Heudorf and Angerer 2001b] as well as in the non-smoking children of smokers [Tsai et al. 2003]. Coal stove exposure or consumption of broiled, fried, or grilled meat contribute to higher levels of 1-hydroxypyrene." |
|
|
|
Post by kammy on Aug 23, 2011 12:51:27 GMT -5
What Is the Biological Fate of the PAHs in the Body? www.atsdr.cdc.gov/csem/csem.asp?csem=13&po=9excerpt "Once PAHs enter the body, several things occur: PAHs are metabolized in a number of organs and excreted in bile and urine PAHs are excreted in breast milk and stored to a limited degree in adipose tissue. Not much data for humans exists regarding the metabolic fate of PAHs. However, information on absorption, distribution, and elimination of these substances is available from animal studies. Pyrene is commonly found in PAH mixtures, and its urinary metabolite, 1-hydroxypyrene, has been used as an indicator of exposure to PAH chemicals. Absorption PAHs are absorbed through ingestion, inhalation, and dermal contact, according to animal study data. The percent absorbed varies in these studies for several reasons, including the vehicle (transport medium) in which the PAHs are found [Kawamura et al. 1988]. In general, PAHs not bound to particulate matter may be absorbed in the lungs better than the same dose found on the surface of airborne particulate matter. Distribution Once absorbed, PAHs enter the lymph, circulate in the blood, and are metabolized primarily in the liver and kidney. PAHs differ with respect to distribution patterns and lipophilic properties [Busbee et al. 1990]. Because of their lipophilic nature, PAHs can accumulate in breast milk and adipose tissue. However, biliary and urinary excretion of PAHs is relatively efficient because of the wide distribution of enzymes that transform PAHs into polar metabolites. Metabolism and Excretion PAHs are predominantly metabolized via CYP enzymes (enzymes in the P-450 mixed-function oxidase system) in the liver [Kapitulnik et al. 1977; Keifer et al. 1988; Monteith et al. 1987]. In addition to the liver and kidneys, metabolism of PAHs occurs in the adrenal glands, testes, thyroid, lungs, skin, sebaceous glands, and small intestines [ATSDR 1995]. PAHs are transformed initially to epoxides, which are converted to dihydrodiol derivatives and phenols. Glucuronide and sulfate conjugates of these metabolites are excreted in the bile and urine. Glutathione conjugates are further metabolized to mercapturic acids in the kidney and are excreted in the urine. The hydroxylated metabolites of the PAHs are excreted in human urine both as free hydroxylated metabolites and as hydroxylated metabolites conjugated to glucuronic acid and sulfate [CDC 2005]. A commonly measured urinary metabolite is 1-hydroxypyrene. Metabolism is a prerequisite for hepatobiliary excretion and elimination through the feces, regardless of route of entry. Key Points Absorption by route varies in animal studies. PAH distribution patterns vary due to differences in lipophilic properties. Metabolism via CYP liver enzymes (enzymes in the P-450 mixed-function oxidase system) is the predominant mechanism of PAH metabolism. PAHs are transformed to epoxides, then to dihydrodiol derivatives and phenols. Excretion occurs via the bile or urine after metabolite conjugation to glucuronides and sulfates. 1- Hydroxypyrene is a commonly measured urine metabolite. |
|
|
|
Post by kammy on Aug 23, 2011 13:17:03 GMT -5
"1-hydroxypyrene, has been used as an indicator of exposure to PAH chemicals." A method to test for PAH in the body: www.umweltdaten.de/gesundheit-e/monitor/PAH-in-urine.pdf"Epoxide hydrolases transform the arene oxides produced during the metabolism of PAH into dihydrodiols, or dihydrodiol epoxides into tetrols. In the course of this process, these enzymes may have toxifying or detoxifying properties. In the so-called phase II metabolism, the epoxides of the PAH are transformed into phenols, diols or tetrols and are conjugated by glutathione transferases, sulfotransferases and DP glucuronyltransferases. They are excreted in the form of ercapturic acids, sulphates and glucuronides in the urine. Concerning the behaviour of PAH in human urinary excretion, most is known about pyrene, whose metabolite, 1-hydroxypyrene, has been susceptible since 1987 to routine testing using a procedure based on high efficiency liquid chromatography [20]. The excretion of 1-hydroxypyrene follows elimination kinetics that are biphasic, with one half-life of between several hours and two days duration, and one half-life of approximately 16 days [21]. One part of the incorporated pyrene is thus swiftly excreted, while the other part is stored in deeper locations, such as the fatty tissue, which it only leaves with a time delay to enter the bloodstream and be excreted. Further studies have confirmed that the first elimination phase for pyrene has half-life durations of between 4 and 35 hours. Sieber et. al.[27] investigated the kinetics of hydroxypyrene excretion in individuals who were occupationally exposed to PAH. Their findings likewise revealed a 2-phase elimination with average half-life durations of approximately 10 hours (6.3 – 15.9 h) and 36 hours (28.8 – 50.1 h). The ultimate carcinogenic compounds in this process are the dihydrodiol epoxides. These may open the oxirane ring and react with the nitrogen bases of DNA by forming covalent bonds. PAH which are classified as carcinogenic vary in their potential to cause cancer according to their chemical structure. Those PAH which have a so-called “bay region”, such as benzo pyrene, display an especially high potential to cause cancer [28]. Most recent findings however show that the cancer-causing effect of the so-called “fjord region PAH”, such as dibenzo[a,l]pyrene, is higher by one or two orders of magnitude. Bay region and fjord region PAH are transformed in the human body into dihydrodiol epoxides, whose epoxide ring is largely protected from attack by the epoxide hydrolases [29] (see Figure 1: bay region; fjord region). These dihydrodiol epoxides are so stable that they can reach the cell nucleus and bind to the DNA there. This bond is seen as the initial step in carcinogenesis [30].
Sources of PAH exposure for humans
The main source of PAH intake is food, on the one hand as a result of airborne PAH precipitating onto cereals, fruit and vegetables, and on the other hand as a result of PAH generated during the preparation of food. For example, smoked food and food grilled on open flames display substantial levels of PAH content [11]. Exposure of humans to PAH from consumption of food containing PAH is discussed in numerous works.
A very important source of PAH exposure among the general population is tobacco smoke [4,37,38]. Smokers’ intake of pyrene in cigarette smoke is of the same order of magnitude as intake from average food consumption [36].
In individual cases, the therapeutic use of ointments and shampoos containing tar may lead to a high additional exposure to PAH [39].
The use of parquet flooring adhesives containing PAH has been identified as a possible source indoors [40,41,42,43]. Parquet adhesives used in residential construction projects up to the 1950’s contained bitumen and tar oils, after which time pure bitumen adhesives were used. Beginning in the middle of the 1970’s, adhesives of different composition were used. The PAH contents of tar oils are considerably higher than that of bitumen.
Human biomonitoring parameters for use in estimating internal exposure / demands on health
In the environment or the workplace, PAH always occur in the form of a mixture that can contain as much as several hundred individual substances. For practical as well as for economic reasons, it is not possible for investigations in the environment or at the workplace to cover and quantitatively determine all PAH individually. This is the reason for selecting a single PAH to represent the whole PAH mixture in an environmental sample, which is subjected to quantitative determination. Traditionally, benzopyrene has been used as the most important PAH indicator in numerous investigations and studies. However, for purposes of quantifying environmental pollution, the United States Environmental Protection Agency (US EPA) recommends using a selection of 16 PAH (see Table 5) [45].
A human biomonitoring investigation (HBM) of PAH is faced with the same difficulties as environmental analyses: it is only possible to investigate a limited number of substances (PAH or their metabolic products) in human body fluids. The selection is made more difficult by the fact that the greater the molecular weight of a PAH, the more it will be excreted in the faeces and thus escape ordinary biological monitoring tests. These are however the PAH with especially high carcinogenic potential.
Hydroxypyrene in urine as a parameter of the internal exposure to PAH
In 1985, Jongeneelen [46] presented for the first time an analytic method (HPLC) which could be used to detect hydroxypyrene, the metabolic product of pyrene, in urine in a relatively simple and reliable manner.
Since then, hydroxypyrene has been measured in numerous occupational medicine and environmental medicine investigations across the world." |
|
|
|
Post by kammy on Aug 23, 2011 14:03:27 GMT -5
Sky, I accidentally typed in 'fungi to PHA' instead of PAH... is it any wonder how the PAH's might be picking up fungi and bacteria? They are experimenting with in the oceans because they have been turned into MAJOR LANDFILLS  to degrade the plastics and other pollution are bacteria and marine fungi that will eat the plastic over time: MARINE FUNGI — DEGRADERS OF POLY-3-HYDROXYALKANOATE BASED PLASTIC MATERIALS www.doiserbia.nb.rs/img/doi/0352-4906/2009/0352-49060916253M.pdf"Today, oceans and estuaries serve as major landfills, and since fungi are an important part of the degrading microbiota, in order to prove their participation in the degradation process, a simple degradation test suitable for fungi and marine conditions had to be developed. Using this medium, 32 strains of marine yeasts and 102 strains of marine mycelial fungi belonging to different systematic and ecological groups were tested for their ability to degrade PHAs" |
|
|
|
Post by kammy on Aug 24, 2011 1:38:43 GMT -5
"Metabolism and Excretion
PAHs are predominantly metabolized via CYP enzymes (enzymes in the P-450 mixed-function oxidase system) in the liver [Kapitulnik et al. 1977; Keifer et al. 1988; Monteith et al. 1987].
In addition to the liver and kidneys, metabolism of PAHs occurs in the adrenal glands, testes, thyroid, lungs, skin, sebaceous glands, and small intestines [ATSDR 1995].
PAHs are transformed initially to epoxides, which are converted to dihydrodiol derivatives and phenols. Glucuronide and sulfate conjugates of these metabolites are excreted in the bile and urine. Glutathione conjugates are further metabolized to mercapturic acids in the kidney and are excreted in the urine."
So, quit smoking, if you do, avoid second-hand smoke. Drink lots of GOOD water. And promote regular bowel movements. The salt water flushes, salt/vit C cleanse, using the epsom salts as a laxative at times...
Q. - CYP enzymes?
|
|
|
|
Post by kammy on Aug 24, 2011 1:46:15 GMT -5
Synonyms for CYP enzymes www.ldc.upenn.edu/mamandel/itre/annotators/cyp/synonyms.html--------------- Cytochrome P450 en.wikipedia.org/wiki/Cytochrome_P450"The cytochrome P450 superfamily (officially abbreviated as CYP) is a large and diverse group of enzymes. The function of most CYP enzymes is to catalyze the oxidation of organic substances. The substrates of CYP enzymes include metabolic intermediates such as lipids and steroidal hormones, as well as xenobiotic substances such as drugs and other toxic chemicals. CYPs are the major enzymes involved in drug metabolism and bioactivation, accounting for ~75% of the total number of different metabolic reactions.[1] The most common reaction catalyzed by cytochromes P450 is a monooxygenase reaction, e.g., insertion of one atom of oxygen into an organic substrate (RH) while the other oxygen atom is reduced to water: RH + O2 + 2H+ + 2e– → ROH + H2O Cytochromes P450 (CYPs) belong to the superfamily of proteins containing a heme cofactor and, therefore, are hemoproteins. CYPs use a variety of small and large molecules as substrates in enzymatic reactions. Often, they form part of multi-component electron transfer chains, called P450-containing systems. Cytochromes P450 have been named on the basis of their cellular (cyto) location and spectrophotometric characteristics (chrome): when the reduced heme iron forms an adduct with CO, P450 enzymes absorb light at wavelengths near 450 nm, identifiable as a characteristic Soret peak. CYP enzymes have been identified in all domains of life, i.e., in animals, plants, fungi, protists, bacteria, archaea, and even viruses." |
|
|
|
Post by kammy on Aug 24, 2011 1:54:05 GMT -5
"insertion of one atom of oxygen into an organic substrate (RH) while the other oxygen atom is reduced to water:
RH + O2 + 2H+ + 2e– → ROH + H2O"
Since we're oxygen deprived such as a cigarette smoker or not getting as much oxygen from our drinking water, plus with the oxygen being depleted in the atmosphere via volcanoes, fires, explosions, off-burning of gases, pollution, etc., etc., common sense might tell us that something in the above 'oxygen needed here' equation is not being carried out?
or/and...
our CYP enzymes are not being allowed to do their job. They have been modified or we have some sort of genetic pre-disposition?
|
|
|
|
Post by kammy on Aug 24, 2011 2:55:26 GMT -5
cont. en.wikipedia.org/wiki/Cytochrome_P450"3. Molecular oxygen binds covalently to the distal axial coordination position of the heme iron. The cysteine ligand is a better electron donor than histidine, which is normally found in heme-containing proteins. As a consequence, the oxygen is activated to a greater extent than in other heme proteins. However, this sometimes allows the iron-oxygen bond to dissociate, causing the so-called "uncoupling reaction", which releases a reactive superoxide radical and interrupts the catalytic cycle. 4. A second electron is transferred via the electron-transport system, from either cytochrome P450 reductase, ferredoxins, or cytochrome b5, reducing the dioxygen adduct to a negatively charged peroxo group. This is a short-lived intermediate state. 5. The peroxo group formed in step 4 is rapidly protonated twice by local transfer from water or from surrounding amino-acid side-chains, releasing one water molecule, and forming a highly reactive species commonly referred to as P450 Compound 1 ( or Compound I). This highly-reactive intermediate was not "seen in action" until 2010,[11] although it had been studied theoretically for many years.[7] P450 Compound 1 is most likely a iron(IV)oxo (or ferryl) species with an additional oxidizing equivalent delocalized over the porphyrin and thiolate ligands. Evidence for the alternative perferryl iron(V)-oxo [7] is lacking. Depending on the substrate and enzyme involved, P450 enzymes can catalyze any of a wide variety of reactions. A hypothetical hydroxylation is shown in this illustration. After the product has been released from the active site, the enzyme returns to its original state, with a water molecule returning to occupy the distal coordination position of the iron nucleus. S: An alternative route for mono-oxygenation is via the "peroxide shunt": Interaction with single-oxygen donors such as peroxides and hypochlorites can lead directly to the formation of the iron-oxo intermediate, allowing the catalytic cycle to be completed without going through steps 3, 4, and 5.[9] A hypothetical peroxide "XOOH" is shown in the diagram. C: If carbon monoxide (CO) binds to reduced P450, the catalytic cycle is interrupted. This reaction yields the classic CO difference spectrum with a maximum at 450 nm. Because most CYPs require a protein partner to deliver one or more electrons to reduce the iron (and eventually molecular oxygen), CYPs are part of P450-containing systems of proteins. Five general schemes are known: CPR/cyb5/P450 systems employed by most eukaryotic microsomal (i.e., not mitochondrial) CYPs involve the reduction of cytochrome P450 reductase (variously CPR, POR, or CYPOR) by NADPH, and the transfer of reducing power as electrons to the CYP. Cytochrome b5 (cyb5) can also contribute reducing power to this system after being reduced by cytochrome b5 reductase (CYB5R). FR/Fd/P450 systems, which are employed by mitochondrial and some bacterial CYPs. CYB5R/cyb5/P450 systems in which both electrons required by the CYP come from cytochrome b5. FMN/Fd/P450 systems originally found in Rhodococcus sp. in which a FMN-domain-containing reductase is fused to the CYP. P450 only systems, which do not require external reducing power. Notable ones include CYP5 (thromboxane synthase), CYP8 (prostacyclin synthase), and CYP74A (allene oxide synthase). P450s in humans Human CYPs are primarily membrane-associated proteins[12] located either in the inner membrane of mitochondria or in the endoplasmic reticulum of cells. CYPs metabolize thousands of endogenous and exogenous chemicals. Some CYPs metabolize only one (or a very few) substrates, such as CYP19 (aromatase), while others may metabolize multiple substrates. Both of these characteristics account for their central importance in medicine. Cytochrome P450 enzymes are present in most tissues of the body, and play important roles in hormone synthesis and breakdown (including estrogen and testosterone synthesis and metabolism), cholesterol synthesis, and vitamin D metabolism. Cytochrome P450 enzymes also function to metabolize potentially toxic compounds, including drugs and products of endogenous metabolism such as bilirubin, principally in the liver. The Human Genome Project has identified 57 human genes coding for the various cytochrome P450 enzymes.[13] Drug metabolism CYPs are the major enzymes involved in drug metabolism, accounting for ~75% of the total metabolism.[1] Most drugs undergo deactivation by CYPs, either directly or by facilitated excretion from the body. Also, many substances are bioactivated by CYPs to form their active compounds. Drug interaction Many drugs may increase or decrease the activity of various CYP isozymes either by inducing the biosynthesis of an isozyme (enzyme induction) or by directly inhibiting the activity of the CYP (enzyme inhibition). This is a major source of adverse drug interactions, since changes in CYP enzyme activity may affect the metabolism and clearance of various drugs. For example, if one drug inhibits the CYP-mediated metabolism of another drug, the second drug may accumulate within the body to toxic levels. CYPs have also been heavily studied in insects, often to understand pesticide resistance. For example, CYP6G1 is linked to insecticide resistance in DDT-resistant Drosophila melanogaster[24] and CYP6Z1 in the mosquito malaria vector Anopheles gambiae is capable of directly metabolizing DDT.[25]" Q. Porphyrin again and porphyria? P450 Compound 1 ?  |
|
|
|
Post by kammy on Aug 24, 2011 2:56:47 GMT -5
cont. en.wikipedia.org/wiki/Cytochrome_P450excerpt "Microbial Microbial cytochromes P450 are often soluble enzymes and are involved in critical metabolic processes. Three examples that have contributed significantly to structural and mechanistic studies are listed here, but many different families exist. Cytochrome P450cam (CYP101) originally from Pseudomonas putida has been used as a model for many cytochromes P450 and was the first cytochrome P450 three-dimensional protein structure solved by X-ray crystallography. This enzyme is part of a camphor-hydroxylating catalytic cycle consisting of two electron transfer steps from putidaredoxin, a 2Fe-2S cluster-containing protein cofactor. Cytochrome P450 eryF (CYP107A1) originally from the actinomycete bacterium Saccharopolyspora erythraea is responsible for the biosynthesis of the antibiotic erythromycin by C6-hydroxylation of the macrolide 6-deoxyerythronolide B. Cytochrome P450 BM3 (CYP102A1) from the soil bacterium Bacillus megaterium catalyzes the NADPH-dependent hydroxylation of several long-chain fatty acids at the ω–1 through ω–3 positions. Unlike almost every other known CYP (except CYP505A1, cytochrome P450 foxy), it constitutes a natural fusion protein between the CYP domain and an electron donating cofactor. Thus, BM3 is potentially very useful in biotechnological applications. Cytochrome P450 119 (CYP119) isolated from the thermophillic archea Sulfolobus acidocaldarius [28] has been used in a variety of mechanistic studies.[11] Because thermophillic enzymes evolved to function at high temperatures, they tend to function more slowly at room temperature (if at all) and are therefore excellent mechanistic models." |
|
|
|
Post by kammy on Aug 24, 2011 2:59:48 GMT -5
This looks like the next stage sphere, Sky - Sulfolobus acidocaldarius en.wikipedia.org/wiki/Sulfolobus"Sulfolobus as a viral host Lysogenic viruses infect Sulfolobus for protection. The viruses cannot survive in the extremely acidic and hot conditions that Sulfolobus lives in, and so the viruses use Sulfolobus as protection against the harsh elements. This relationship allows the virus to replicate inside the archaea without being destroyed by the environment." The Sulfolobus is contained inside the hydrocarbon, the 'others' are contained inside the Sulfolobus. |
|
|
|
Post by kammy on Aug 24, 2011 6:59:31 GMT -5
The next layer of attack: This second sphere seen that is predominately seen in all human samples is formed from the 'hydrocarbon' sphere, the black carbon sphere turns more into this brown, sometimes reddish sphere; human sample, 100x, the carbon sphere turning into the brown sphere:  human sample 450x below. Notice the strong green halo, I think is indicative of iron which I stated 3 years ago:  This second appearing sphere cannot be just Sulfolobus acidocaldarius by itself, it is some sort of combination that I have documented over time that show its many different properties and described them; turns into a white 'fungus', a black one, one contains a baculoviral element... etc., all are able to be held inside the 'hydrocarbon' sphere, eventually show themselves as this secondary sphere when cultured for any length of time, I think up to within 1 day they can appear, I'd have to look at my notes. Which all seemingly look the same but it has many different characteristics, too many different ways it's preforming. It seems to me that anything that is being added to the oceans is ending up inside of us. I also see the calcite crystals, as per Marc Darrah's findings, in a stacked formation that are formed from the baculoviral aspect of one of these secondary spheres as occlusion bodies. Calcite is being added to the oceans in the form of chalk. It sounds like and with just a basic visual inspection - could be a combination of the 5 mentioned above and then with possibly 'others'. When I look at the basic stock photos of these below, I can see some aspect about each of them that I can match with photographs: Cytochrome P450 enzymes Sulfolobus acidocaldarius Pseudomonas putida Saccharopolyspora erythraea Bacillus megaterium I believe you mentioned Staninger, Kolb, or Grady in a post where they thought Pseudomonas putida was involved. Page 11 - that pyryll was used in the Corexit........so it came from DOE.
So, back to our lovely bucky ball cages. idea of Teller worked for Energy, physics. atomic bomb. = ===============================\
DOE created the microbes.
One of the thing they did was the p. putida formation from p auringinosa when hit the oil it formed
balls, what is happening now in gulf, these do not go away, they have been used before. Roberta L
in New Zeeland, was hit with this, grady I bel was too.
Have your read this on LB? or do you all remember this?=================
A Theory - Pseudomonas Putida = Root Cause
Post by grady on Jan 5, 2008, 10:11pmI have spoken with Dr. Staninger and Dr. Kolb both on the phone. Dr. Staninger stated that she has proven that our bodies are creating plasticines (spelling) or in laymans terms a type of platic in our bodies and also tubules (carbon I think, hollow tubes) but I cannot remember for sure if she said carbon, however, she did talk alot about our body creating plastic.Now, when I spoke to Dr. Kolb (she is out of Atlanta and has met with the CDC on this I am told) she stated she doesn't know for sure what this is but s trongly suspects bacteria. She asked me if I had been to the beach and dug in the sand? I said yes, we take shovels and build massive sand castles. She stated that so many people that she is treating answer that question the same way it cannot be coincidence. The subject of bacteria, the same kind brewed to eat oil spills came up, she said to avoid plastics, food that comes in plastics and so on.I do not think these two doctors are even aware of each others work frankly. Well, tonight I did some research, look what I found.
Bacteria Eating Up Oil Spills and Producing Biodegradable Plastic - And could also offer a new solution for plastic recyclingBacteria are everywhere and are capable of both highly useful and highly disastrous effects. They are also capable of living in the outmost unfavorable conditions. Biotechnologists have long tried to design new types of bacteria capable of digesting pollution or various artificial non-biodegradable products.
This would be the BAC, bacterial artificial chromosomes.Now, biologists from the University College Dublin in Ireland have found that a strain of Pseudomonas putida can not only thrive in an environment that most creatures would consider deadly but can also produce useful byproducts.Kevin O'Connor and his colleagues produced a chemical ocktail made up of more than 80 percent styrene oil plus low volumes of other toxicants. Frankly, they haven’t expected that their test bacteria, P. putida CA-3, a special strain of a common soil microbe, would do too well. However, the bacteria managed to turn 64 grams of undistilled styrene oil into nearly 3 grams of additional bacteria. In the process, the bacteria also produced 1.6 grams of a biodegradable plastic called polyhydroxyalkanoates, or PHA. The PHA is already used to make everything from forks to vitamins.
We cannot yet say that this bacterium literally changes an environmental problem into a beneficial substance because it also produces some amounts of other toxic byproducts such as toluene. Nevertheless, it is a good start. The chemicals the bacteria likes can be produced from polystyrene through a process called pyrolysis. Thus, they may be useful in recycling plastics. "Due to the general applicability of pyrolysis for plastic conversion to an oil and the large number of microorganisms capable of PHA accumulation from a vast array of molecules, the principle of the process described here can be applied for the recycling of any petrochemical plastic waste," the scientists write.
A bacteria (found not created) that comes from the soil can turn chemicals into plastic, well I'll be darned, who'd a thought, any takers ... Honestly, I really thing this is adding up ... I know there is a fungal component, but perhaps it's because our skin is riddled with holes and such from the plastic fibers that are being produced, the fibers are the result of the bacterial process going on in our bodies, the fungal spores get in there only because of these perfurations in our skin. Please excuse the egotistical title :PRe: Grady's new Theory is Brilliant Post by grady on Jan 5, 2008, 10:20pm Futher more, The article talks of Toluene as a toxic by product when the bacteria creates plastics from as the article states "a vast array of molecules" (which I am sure can be found in our bodies) it creates Toluene, this seems VERY SIMILAR to the goop folks talk about, I remember just the other day someone talking about this clear stickly liquid, guys, this is our baby, I know this is it.Look what it says about Toluene en.wikipedia.org/wiki/TolueneToluene may enter the human system not only through vapour inhalation from the liquid evaporation, but also following soil contamination events, where human contact with soil, ingestion of contaminated groundwater or soil vapour off-gassing can occur.The toxicity of toluene can be explained mostly by its metabolism. As toluene has very low water solubility, it cannot exit the body via the normal routes (urine, feces, or sweat).[citation needed] It must be metabolized in order to be excreted. The methyl group of toluene is more easily oxidized by cytochrome P450 than the benzene ring. IMPORTANTTherefore, in the metabolism of toluene, 95% is oxidized to become benzyl alcohol.[4] The toxic metabolites are created by the remaining 5% that are oxidized to benzaldehyde and cresols.[5][6] Most of the reactive products are detoxified by conjugation to glutathione but the remainder may severely damage cells.[7] Re: Grady's new Theory is Brilliant Post by grady on Jan 5, 2008, 10:34pm I'll continue. I've always rejected the plastic theory and always wondered about these so called "fibers" that won't burn. I've seen them under my skin like a mesh (not so bad now) and wondered how I could live with this stuff all throughout my body? If it would never break down or degrade how could I continue and like this and not die? Well, the article states that the plastic is "biodegradable plastic called polyhydroxyalkanoates" and breaks down in about 55 days. This is all clicking folks, CLICK, CLICK, CLICK. Let me summarize: ( This is grady's summary) 1) Something akin to "Pseudomonas putida" has gotten into our systems. 2) It is producing plastic fibers and tubules as a by product of its life process 3) It is also producting "toluene" which we need to get rid of via "glutathione " (more on that in a follow up post tomorrow) 4) No doubt, the toxins given off by any bacteria can make one feel ill by i ncreasing the toxic burden on the body and toluene is not the only toxic by product I'm sure.5) The plastic fibers (others have had hard chunks of plastic come right out of thier skin and lips) purferate the skin making for perfect little entry points for fungal sporesIt could be this soil bacteria is even attractive to bugs/worms, who knows, kill the bacteria, slay the beast. Perhaps that is why anti-biotics are working so well for some, we just need the right killer. Re: Grady's new Theory is Brilliant Post by skytroll on Jan 6, 2008, 1:52am Grady, I believe that toulene is in chemtrails also.So check out polyonucleotides, artificial DNA.
Going into detail over here: tinyurl.com/3ytgnrmorgellonsgroup.proboards23.com/index.cgi?action=display&board=nanodiscuss&thread=1199169704&page=1 Then check out "cold fusion" and the Sigma Aldrich DendrimersWill put that link back up. Skytroll lymebusters.proboards.com/index.cgi?board=rash&action=print&thread=7441==================== Skyship ====================================== ================== |
|
|
|
Post by kammy on Aug 24, 2011 7:13:47 GMT -5
|
|
|
|
Post by kammy on Aug 24, 2011 7:45:58 GMT -5
Ok, we've given a definition of the first level PAH hydrocarbon sphere which took us into the human enzymes involved in dealing with hydrocarbons in our bodies. I believe we're getting a handle on the next layer of attack, the secondary sphere.
This thread is dedicated to the 'carbon ball' sphere. These other findings should probably have their own threads? I am currently working on the particulate matter aspect in a thread by itself. It seems that the PAH hydrocarbon has a friend - particulate matter, in fact - that it has a lot of 'friends'.
|
|
|
|
Post by kammy on Aug 27, 2011 6:35:47 GMT -5
A Looming Oxygen Crisis and Its Impact on World’s Oceans 05 AUG 2010: ANALYSIS e360.yale.edu/content/feature.msp?id=2301excerpt "As ocean currents oscillate naturally, upwellings of deep-ocean water can deliver nutrients to coastal waters, triggering an explosion of growth and driving down oxygen levels. Volcanoes can alter oxygen levels, too, by creating a haze that blocks sunlight, thus temporarily cooling the ocean’s surface and allowing more oxygen to dissolve into the water. In recent years some worrying signals have started to emerge from the noise. In 2006, for example, oxygen levels off the coast of Oregon dropped to record lows. Reefs that had been packed with rockfish and other animals suddenly became ecological ghost towns. A drop in oxygen may also cause the ocean's bacteria to change. Bacteria that need oxygen will no longer be able to thrive in oxygen-free zones of the ocean. But these dead zones will foster the growth of many species of bacteria for whom oxygen is toxic. Some of these oxygen-hating microbes produce nitrogen compounds that are among the most potent greenhouse gases ever measured. " |
|
|
|
Post by skyship on Aug 29, 2011 0:13:25 GMT -5
One thing of greatest importance we have not looked at entirely, is nitrogen. Its relationship to amyloid: "Prion Domain Initiation of Amyloid Formation in Vitro from Native Ure2p 2. 2 Laboratory of Structural Biology, National Institute of Arthritis and Musculoskeletal and Skin Diseases, National Institutes of Health, Bethesda, MD 20892–2717, USA. 3. 3 Department of Biochemistry and Molecular Biology, Uniformed Services University for Health Sciences, Bethesda, MD 20814, USA. Abstract The [URE3] non-Mendelian genetic element of Saccharomyces cerevisiae is an infectious protein (prion) form of Ure2p, a regulator of nitrogen catabolism. Here, synthetic Ure2p1−65 were shown to polymerize to form filaments 40 to 45 angstroms in diameter with more than 60 percent β sheet. Ure2p1−65 specifically induced full-length native Ure2p to copolymerize under conditions where native Ure2p alone did not polymerize. Like Ure2p in extracts of [URE3] strains, these 180- to 220-angstrom-diameter filaments were protease resistant. The Ure2p1−65-Ure2p cofilaments could seed polymerization of native Ure2p to form thicker, less regular filaments. All filaments stained with Congo Red to produce the green birefringence typical of amyloid. This self-propagating amyloid formation can explain the properties of [URE3].".............. www.sciencemag.org/content/283/5406/1339.short====================== Homosapiens: Molecular pathogens, so many "little bodies", inclusion bodies etc. Many amyloid related. Hardending of arteries, skin, etc................the "lizard skin" appearance. www6.ufrgs.br/favet/imunovet/molecular_immunology/pathohomoprocess_regressive.htmlSkyship |
|
|
|
Post by kritters on Aug 29, 2011 18:42:46 GMT -5
|
|
|
|
Post by skyship on Nov 15, 2011 5:07:56 GMT -5
Alright - let me look at hypha and cellulose micelles as per post 93 to go by: reference.findtarget.com/search/biopolymer/"Biopolymers are polymers produced by living organisms. Cellulose, starch, chitin, proteins, peptides, DNA and RNA are all examples of biopolymers, in which the monomeric units, respectively, are sugars, amino acids, and nucleotides.Cellulose is both the most common biopolymer and the most common organic compound on Earth. About 33 percent of all plant matter is cellulose. E.G. The cellulose content of cotton is ~ 90 percent and that of wood is ~ 50 percent. 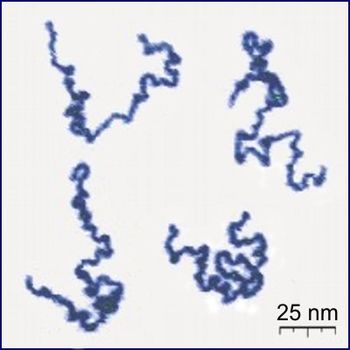  Cellulose Many polymer researchers believe that polymer chemistry had its origins with the characterization of cellulose. Cellulose was isolated for the first time some 150 years ago. Cellulose differs in some respects from other polysaccharides produced by plants, the molecular chain being very long and consisting of one repeating unit (cellobiose). Naturally, it occurs in a crystalline state. From the cell walls, cellulose is isolated in microfibrils by chemical extraction. In all forms, cellulose is a very highly crystalline, high molecular weight polymer, which is infusible and insoluble in all but the most aggressive, hydrogen bond-breaking solvents. Because of its infusibility and insolubility, cellulose is usually converted into derivatives to make it more user friendly in processing. Some fungi can secrete enzymes that catalyze oxidation reactions of either cellulose itself or the lower molecular weight oligomers produced from the enzymatic hydrolysis of cellulose. Of these, the peroxidases can provide hydrogen peroxide for free radical attack on the C2–C3 positions of cellulose to form ‘aldehyde' cellulose, which is very reactive and can hydrolyze to form lower molecular weight fragments. Bacteria also secrete enzymes, some of which form complexes that act jointly in degrading cellulose to form carbohydrate nutrients which the microorganisms utilize for survival." |
|