|
|
Post by lilsissy on Dec 7, 2010 10:10:50 GMT -5
I have been chasing my tail on this thread but now I see where you are going, very interesting indeed. Off topic here but fits in the general topic of earth cahnges. howsthatpossible.com/ |
|
|
|
Post by lilsissy on Dec 7, 2010 10:18:17 GMT -5
Wonder the DMS , does it tie in with the marine viruses? lymebusters.proboards.com/index.cgi?board=rash&action=display&thread=13354also first comes to mind the Chitin, breaks open our cells by skin contact, our White Blood Cells form pseudopodia to reach out to accept the chitin. This forms a gel and the chitin is literally pulled into the cell. Poly N-AG Remember the patent I called the IT patent. That was the component that broke open the cells. Wonder how it ties in also reading this www.morgboard.proboards.com/index.cgi?board=general&action=display&thread=503&page=5I believe we all feel it is fungal related, yes! The two things that stand out to me is this.. Nano's will just be spontaneous unorganized unless scattered on a fugal mat. Researchers use fungus to align them. Alginate was used to align the PEDOT for an artificial nervous system . Their are different recipes for Alginates. University of Michigan received the acclaim for their Alginate. Wonder if we can find thier patent? |
|
|
|
Post by kammy on Dec 7, 2010 10:56:08 GMT -5
Kammy , I am reading all of this but I must admit it all makes my head spin. I have read these debates some in the past but I must admit I have decided very little on the subject. It is like falling into another bottomless pit trying to figure out the truth of the climate change debate. I do feel that I know that a group are killing our Scientist , who , why they are doing this I do not know. The other day I spend an hour or so looking over the research activities of the Dead Scientists, I do not believe that all where killed. One thing was certain ,way to many of these Scientist had previously been studying Mad Cow Disease...prions... What I feel is occurring is that the United States has put up a nano shield that can be assembled to meet incoming rockets. It is thick like pea soup ,when the rocket hits it at a high rate of speed it is like hitting a brick wall. These nano's float in the upper atmosphere independent from one another until a catalyst causes then to form the pea soup phase. This was what I remember from the little research myself and my sister Karen had done a few years ago. I think maybe Sky was in on it too. The unfortunate thing is that Our Government had a call out to make safer for humans nano's for this program. Also I believe that this upper atmosphere conglomeration of nano's carry fungus. There was a woman who received acclaim for finding that forest fires carried fungi across the Atlantic piggybacked on the carbon from the fires. Grass when burnt at high temps creates natural nano's. So if our Government is spraying the atmosphere ( we had articles to prove that was occurring) for either climate control or for Star War Missile Shield Programs , they may have inadvertently caused a scenario foretold, The sun will not give off her light as foretold by John in the book Revelation. Large fires are also foretold . I personally feel Atmospheric heaters such as HAARP are being used as weapons. but I have found evidence that also makes me believe that beams of light collected on a prism can be directed at a target and cause spontaneous fires to occur at once. well I'll see if I can find the article for you. Did you ever read of the Italy fires that our Govenrmnet said was caused by forces not of this earth ? Hi Sis, I just now saw this post. I know that controversy is tedious to deal with, I had to set up some history though, around how everything is coming together. Of course, there's going to be a lot of controversy around our mystery disease, we've already had wind of how they operate: they fund one group to take a certain stance, they turn around and fund an opposing group so that their will be dissension and confusion around the subject - we have to look past the smoke and mirrors. We need to get educated pronto, for whatever reason, most of us don't seem to care too much about this issue, why? - it's out of our hands, it's too big, we don't have good information, the information is confusing... only a super power can come in and fix this... just what they want. They are using human carbon emissions as the reason to justify de-population, they are most likely going to use biologicals, such as what has happened to us, as their de-population mechanism as has been published. It has been stated that they are cheap to produce and if you use something that is already there in predominance in nature to tag along with...? I believe we are finding what is at the center of our disease and its main components. We will find that our sphere has properties LIKE an absorbent sponge that picks up anything it comes into contact with and will tie in with all our other research, the things you mentioned. I just showed how our fibers are most likely being created, little baby steps to understanding, hang in there. |
|
|
|
Post by kammy on Dec 7, 2010 11:00:21 GMT -5
I have been chasing my tail on this thread but now I see where you are going, very interesting indeed. Yes, I wanted to jump right to the 'meat' but thought... I don't understand this issue or haven't followed it and it won't hurt to know - afterall, it affects us all. Boy - does it ever! By clicking on another link to a post you made, Sis - I came on that new microscope article that showed me what I've been looking for all along... that lead to this. See how God works in mysterious ways?... we're all a part of this. |
|
|
|
Post by katinka on Dec 7, 2010 11:02:08 GMT -5
This picture explains exactly what's happening in our tropospheric atmosphere: saga.pmel.noaa.gov/review/dms_climate.html Oceanic dimethylsulfide (DMS) is the major natural source of sulfur to the atmosphere and contributes both to the tropospheric sulfur burden and to particle formation and growth in the atmosphere. The atmospheric sulfate aerosol particles that evolve from biogenically-derived DMS emissions play a role in the global radiation balance directly through the upward scatter of solar radiation and indirectly as cloud condensation nuclei. |
|
|
|
Post by kammy on Dec 7, 2010 11:05:44 GMT -5
"Wonder the DMS , does it tie in with the marine viruses?"
Yes, I just read something about that last night... yes, unfortunately, EVERYTHING will tie in.
|
|
|
|
Post by kammy on Dec 7, 2010 11:20:53 GMT -5
Off subject, Jen - that is interesting!, it looks like our 'fungus' !!!! howsthatpossible.com/wp-content/uploads/2010/11/antimatter.jpgSo... how did they get this 'stuff' to form - by banging certain particles together. So, our spheres and 'fungus' are the closest to "god energy" that man can get? ------------------ I'm thinking - we probably have these DMS micelles inside of us, in our intestines?... they appear to be naturally in our food...? so, are they a part of us naturally? It's only when we get an overload of free radicals and these micelles that we show symptoms?... That by reducing our free radical load - we can stop the formation of fibers?... what brings on apoptosis of the spheres, though? I'm just the pointer... you smarter gals need to figure out what's going on inside the body. |
|
|
|
Post by katinka on Dec 7, 2010 11:24:39 GMT -5
So, either the micelles can do this naturally or with the addition of something to cause this to happen, I would think - it is the addition of something since this our disease is a new phenomena. It has been compared to a free radical polymerization chain reaction. Contact with some sort of free radical catalyst?
All transition metals, with the exception of copper contain one electron in their outermost shell and can be considered free radicals. Copper has a full outer shell, but loses and gains electrons very easily making itself a free radical. In addition iron has the ability to gain and lose electrons (i.e. (Fe2+«Fe3+) very easily.
This property makes iron and copper two common catalysts of oxidation reactions. Iron is major component of red blood cells (RBC). A possible hypothesis is that the stress encountered during may break down RBC releasing free iron. The release of iron can be detrimental to cellular membranes because of the pro-oxidation effects it can have. Zinc only exists in one valence (Zn2+) and does not catalyze free radical formation. Zinc may actually act to stop radical formation by displacing those metals that do have more than one valence.It comes to the so called Fenton's reaction. The production of Hydroxyl radicals...later cell injury and apoptosis. Carnicom has also shown this in his latest experiments.  Like I stated before I believe it is very important for us to antagonize iron and copper with high doses of zinc to stop or at least slow down this process. Magnesium to relieve stress symptoms. |
|
|
|
Post by kammy on Dec 7, 2010 11:43:28 GMT -5
"Like I stated before I believe it is very important for us to antagonize iron and copper with high doses of zinc to stop or at least slow down this process.
Magnesium to relieve stress symptoms."
Very good and sound advice, Kat - I'm sure you're on the right track. This should slow down fiber formation - but what will slow down the sphere reproduction?
Add to this sphere - a protective biofilm. Sometimes the sphere is inside the biofilm... all the artifacts can be inside the biofilm, for that matter.
To keep it simple for now - Let's say the biofilm is an after production of when the sphere has matured... so if the sphere never matures - it should cut down on the biofilm too.
We've got to figure out how to cause apoptosis of these DMS(P) micelles or what have most likely morphed into tropospheric particles?
|
|
|
|
Post by katinka on Dec 7, 2010 11:44:12 GMT -5
Tropospheric particles: Tropospheric air pollution has impacts on scales ranging from local to global. Reactive intermediates in the oxidation of mixtures of volatile organic compounds ( VOCs) and oxides of nitrogen (NOx) play central roles: the hydroxyl radical (OH), during the day; the nitrate radical (NO3), at night; and ozone (O3), which contributes during the day and night. Halogen atoms can also play a role during the day. Here the implications of the complex VOC-NOx chemistry forO3 control are discussed. In addition, OH,NO3, andO3 are shown
to play a central role in the formation and fate of airborne toxic chemicals, mutagenic polycyclic aromatic hydrocarbons, and fine particles.virtual.yosemite.cc.ca.us/scroggins/SciiMag/TroposphericAi%20Pollution97.pdf |
|
|
|
Post by lilsissy on Dec 7, 2010 11:55:17 GMT -5
What I know about D.M.S.O. is very little but If you add 3 drops in your drinking water it helps to unfold Prions. Looking into it Kam, D.M.S.O. also opens up membrane barriers. Sulfur centers memtioned here, I am intolerant to sulphur so is Karen I believe we may have Porphyria Almost certain, it is an abnormal breaking open of red blood cells. Sulfphur is contradicted in those who have Phorphria In fact Morgellons itself has much in common with Porphyria. Facts on D.M.S.O. www.absoluteastronomy.com/topics/Dimethyl_sulfoxideI can see where this would be usefull for giving pathogens by air contact and I am wondering if it is not the very element that allows chitin , Poly N- AG to open our white blood cells, Marine biology The It patent was owned by a Maribe Biology Co. Brain Scientists study Marine Biology extensively. www.absoluteastronomy.com/topics/Dimethyl_sulfoxidev |
|
|
|
Post by kammy on Dec 7, 2010 11:58:48 GMT -5
Apoptosis of Tropospheric particles or DMS(P)...? worldwidescience.org/topicpages/a/aerosol+mass+spectrometry.html"The behavior of small aerosol particles depends on a number of their physical and chemical properties, many of which are strongly coupled. The size, internal composition, density, shape, morphology, hygroscopicity, index of refraction, activity as cloud condensation nuclei and ice nuclei, and other attributes of individual particles - all play a role in determining particle properties and their impacts. The traditional particle characterization approaches rely on separate parallel measurements that average over an ensemble of particles of different sizes and/or compositions and later attempt to draw correlations between them. As a result such studies overlook critical differences between particles and bulk and miss the fact that individual particles often exhibit major differences." Keep looking... |
|
|
|
Post by kammy on Dec 7, 2010 12:16:37 GMT -5
BINGO! Great article, Sky - this photo is the life cycle that's probably now been altered from whatever is being added... but close enough for us to get an idea: 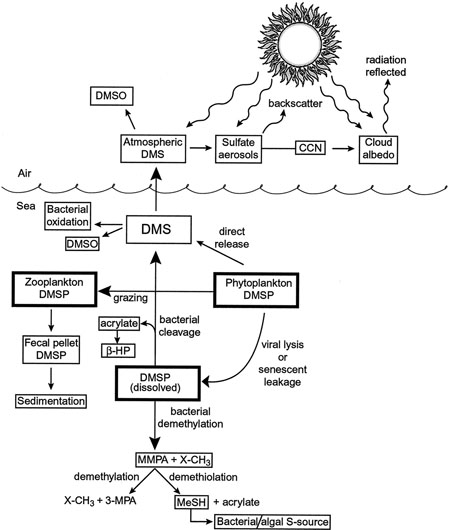 Ok, this diagram gives us a BINGO... you see 'fecal pellet'? I have already shown in the life cycle where the insect is 'pooping' the next generation with a 'carbon ball' !! This carbon ball appears to be DMSP. tinypic.com/r/fdxcus/7Look at post 41, the chart there is what used to be happening... lol we're not looking for DMSO, necessarily... I changed it to show the insect pooping the DMSP... the tinypic at the bottom left. We're looking for what DMSP can turn into as the possible CARRIER, SPHERE that is holding and hosting our disease, the Pac-Man dot eater... |
|
|
|
Post by lilsissy on Dec 7, 2010 12:23:18 GMT -5
|
|
|
|
Post by lilsissy on Dec 7, 2010 12:33:12 GMT -5
|
|
|
|
Post by katinka on Dec 7, 2010 13:15:31 GMT -5
en.wikipedia.org/wiki/HalogenHalogen atoms: comprising fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At) Halogens are highly reactive, and as such can be harmful or lethal to biological organisms in sufficient quantities. This high reactivity is due to the atoms being highly electronegative due to their high effective nuclear charge. They can gain an electron by reacting with atoms of other elements. Fluorine is one of the most reactive elements in existence.
|
|
|
|
Post by kammy on Dec 7, 2010 13:30:03 GMT -5
|
|
|
|
Post by kammy on Dec 7, 2010 13:57:07 GMT -5
Why Are Cells Powered by Proton Gradients? www.nature.com/scitable/topicpage/why-are-cells-powered-by-proton-gradients-14373960"Why do virtually all cells "breathe" by pumping protons (hydrogen ions) across a membrane? Back in the 1940s, Efraim Racker had just figured out the mechanism by which cells glean a little energy from the breakdown of glucose in the absence of oxygen, a pathway known as glycolysis. In glycolysis, phosphate groups are transferred directly from sugar molecules onto ADP to form ATP. The whole pathway is pure chemistry, involving the reaction of one molecule with another, and therefore obeys the laws of stoichiometry; that is, you can balance the equations. But one glaring problem with aerobic respiration is that it doesn't balance. Exactly how much ATP is produced per oxygen molecule consumed? The amount varies, but it's somewhere around 2.5 ATP molecules. That works out to 28–38 ATPs per glucose — again, a variable number, and never an integer (Silverstein 2005). Aerobic respiration is not stoichiometric, so it's really not chemistry. And that's why the long search for a high-energy chemical intermediate (a molecule able to transfer the energy from the oxidation of glucose to form ATP) was doomed to failure. In place of such an intermediate, Mitchell proposed a proton gradient across a membrane: the proton motive force (Mitchell 1961). It works much like a hydroelectric dam. The energy released by the oxidation of food (via a series of steps) is used to pump protons across a membrane — the dam — creating, in effect, a proton reservoir on one side of the membrane. The flow of protons through amazing protein turbines embedded in this membrane powers the synthesis of ATP in much the same way that the flow of water through mechanized turbines generates electricity. This explains why respiration is not stoichiometric: a gradient, by its very nature, is composed of gradations. That begs the question, how did mitochondria get inside an archaeon? Summary Peter Mitchell's demonstration that ATP synthesis is powered by proton gradients was one of the most counterintuitive discoveries in biology, and it took a long time to be accepted. The precise mechanisms by which a proton gradient is formed and coupled to ATP synthesis (chemiosmotic coupling) is now known in atomic detail, but the broader question that drove Mitchell — why are proton gradients so central to life? — is still little explored. Recent research suggests that proton gradients are strictly necessary to the origin of life and highlights the geological setting in which natural gradients form across membranes, in much the same way as they do in cells. But the dependence of life on proton gradients might also have prevented the evolution of life beyond the prokaryotic level of complexity, until the unique chimeric origin of the eukaryotic cell overcame this obstacle." |
|
|
|
Post by kammy on Dec 7, 2010 14:56:11 GMT -5
This abstract shows the requirements for a good chain polymerization in DMSP: pubs.acs.org/doi/abs/10.1021/ma991665s"Provided that the terminal hydroxyl groups of the PDMS macroinitiators are carefully titrated to the corresponding potassium alcoholates, reasonably good control over the DMA chain length was achieved." |
|
|
|
Post by kammy on Dec 7, 2010 15:16:54 GMT -5
They are carriers! Shows that micelles are found naturally in the human body: en.wikibooks.org/wiki/Structural_Biochemistry/Lipids/Micelles"Micelles are also at work in the human body. Micelles help the body absorb lipid and fat soluble vitamins. They help the small intestine to absorb essential lipids and vitamins from the liver and gall bladder. They also carry complex lipids such as lecithin and lipid soluble vitamins (A, D, E and K) to the small intestine. Without micelles, these vitamins will not be able to be absorbed into the body which will lead to serious complications. Micelles also help clean the skin. Many facial washes use micelles to perform this task. They clean the skin by removing oil and other substances without the need of being washed afterward." Vesicles are also found in the human body: "A vesicle is a intracellular membrane bound sac that transports and stores substances within the cell. These vesicles store, transport, and digest waste and products from the cell. They can fuse with the plasma membrane to release things from the cell or come into the cell and put things in. Vesicles are important since they play a role in metabolism, transport, enzyme storage, and are chemical reaction chambers." |
|
|
|
Post by kammy on Dec 7, 2010 15:37:10 GMT -5
Emulsification, Hydrolysis and Micelle Formation in human body www.vivo.colostate.edu/hbooks/pathphys/digestion/smallgut/absorb_lipids.html"As monoglycerides and fatty acids are liberated through the action of lipase, they retain their association with bile acids and complex with other lipids to form structures called micelles. Micelles are essentially small aggregates (4-8 nm in diameter) of mixed lipids and bile acids suspended within the ingesta. As the ingesta is mixed, micelles bump into the brush border of small intestinal enterocytes, and the lipids, including monoglyceride and fatty acids, are taken up into the epithelial cells. Once inside the enterocyte, fatty acids and monoglyceride are transported into the endoplasmic reticulum, where they are used to synthesize triglyeride. Beginning in the endoplasmic reticulum and continuing in the Golgi, triglyceride is packaged with cholesterol, lipoproteins and other lipids into particles called chylomicrons. Remember where this is occurring - in the absorptive enterocyte of the small intestine. Chylomicrons are extruded from the Golgi into exocytotic vesicles, which are transported to the basolateral aspect of the enterocyte. The vesicles fuse with the plasma membrane and undergo exocytosis, dumping the chylomicrons into the space outside the cells. Because chylomicrons are particles, virtually all steps in this pathway can be visualized using an electron microscope, as the montage of images to the right demonstrates." 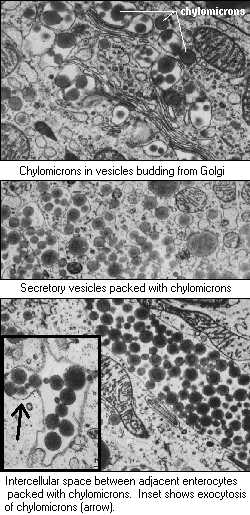 |
|
|
|
Post by kammy on Dec 7, 2010 16:31:18 GMT -5
Micelles found naturally in food? www.foodylife.com/food-safety-and-hygiene/603/molds-in-foods/26 Apr, 2009 Molds In Foods "But if there is mold grown in food, micelles penetrate the inside of the food. If mold produce mycotoxin, toxin generally exist in and around micelles. In some cases, toxin may spread whole food." Dairy Chemistry and Physics www.foodsci.uoguelph.ca/dairyedu/chem.html *I have photographs of milk also. en.wikipedia.org/wiki/Casein"It is found in milk as a suspension of particles called casein micelles which show some resemblance with surfactant-type micellae in a sense that the hydrophilic parts reside at the surface. The caseins in the micelles are held together by calcium ions and hydrophobic interactions. There are several models that account for the special conformation of casein in the micelles (Dalgleish, 1998). One of them proposes that the micellar nucleus is formed by several submicelles, the periphery consisting of microvellosities of κ-casein (Walstra, 1979; Lucey, 2002). Another model suggests that the nucleus is formed by casein-interlinked fibrils (Holt, 1992). Finally, the most recent model (Horne, 1998) proposes a double link among the caseins for gelling to take place. All 3 models consider micelles as colloidal particles formed by casein aggregates wrapped up in soluble κ-casein molecules." |
|
|
|
Post by kammy on Dec 7, 2010 17:25:30 GMT -5
Backing up... I'm thinking that when certain factors are added to the oceans such as volcanic ash, petroleum products, forest fires, chemtrail emissions, we can assume that there are changes to the stratosphere and the troposphere's particles. These particles come back down in rain and can penetrate the food, get in the water supply, etc. I'm looking at tropospheric particles penetrating food. I can see an old report from 1958 when they studied the effects on the plants and residents of certain areas after the particles got into the food chain. This is called 'tropospheric fall-out', when it happens around the world, it is called 'global fallout': Here's an old paper from 1958 to show how long they've been studying this... www.unscear.org/docs/reports/1958,%2013th%20session%20(Suppl.%20No.17)/1958final-1_unscear.pdf |
|
|
|
Post by lilsissy on Dec 7, 2010 17:49:29 GMT -5
|
|
|
|
Post by kammy on Dec 8, 2010 11:44:43 GMT -5
I'm thinking... here's human blood on a slide, self-diagnosed Morgellons patient, 100x: tinypic.com/r/v4s3k0/7You see the micelle in the blood? And then there are particles in the blood: tinypic.com/r/mtni3t/7Strange formations: tinypic.com/r/123bjhk/7And, 'stringy' blood, like a pollen tube is forming? : tinypic.com/r/wtimnm/7I'm sure if I look, I see these micelles in our saliva, in our urine, in a cheek scraping, in places that have liquid. There's particles and spheres coming out of my skin. These are being produced internally not necessarily at the site of the lesion, my body has chosen certain sites to rid these causing lesions. There is 'machinery' that's gone awry inside of me - it is a micelle to fiber machine. I believe there's a point in their development in which they are very, very small. Possibly, my microscope can't even pick them up in this state? You see how small the cloud condensed nucleus are? This big sphere at the bottom represents a typical rain drop:  Cloud condensed nucleus should be a precursor form of the micelle that is inside of us? I'm trying to identify how DMS(P) micelles that are modified and are in the troposphere and come back down into the food and water chain, then we eat, drink and breathe them. They meet all the characteristics of what we're looking for. Inside the human body, in the intestines - something is happening with these micelles in our gut linings, in the chemical mix of the unnatural with what our body does naturally... something at this point is going wrong and is the place to look to find what is causing us to be evidently sick. That possibly a gastroenterologist might be a doctor to approach with looking at what is happening with our Chylomicron process? What test would that be? |
|
|
|
Post by kammy on Dec 8, 2010 12:08:32 GMT -5
This abstract is over my head, the images at Figure 3 are very similar to typical Morgellons, Figure 2 is comparable to the photo I posted above of the particles in blood: Fast Release of Lipophilic Agents from Circulating PEG-PDLLA Micelles Revealed by in ViWo Fo¨rster Resonance Energy Transfer Imaging www.kinam.com/Articles/PMicelles%20Papers/Chen%2008%20Langmuir.pdfand they are calling the micelles in Figure 3 - 'Fret' micelles. Ok, for those of you with medical knowledge... some areas to look in?: Fret micelles Interaction of lipid micelles with blood serum lipoproteins Dysfunctional HDL Cholesterol micelles Ferritin and Ferruginous Micelles in Normal Erythroblasts We need to figure out how do the micelles from the ocean that get into the clouds... that end up in our bodies - where do they go in the human body and what do they do there? lol I'm just assuming they congregate first in the intestines and lungs. ?? |
|
|
|
Post by kammy on Dec 8, 2010 13:31:53 GMT -5
Looking at the definition of 'fallout', it has mostly been associated with radioactive particles in the past. encyclopedia2.thefreedictionary.com/fallout"fallout, minute particles of radioactive material produced by nuclear explosions (see atomic bomb; hydrogen bomb; Chernobyl) or by discharge from nuclear-power or atomic installations and scattered throughout the earth's atmosphere by winds and convection currents. Heavier fallout particles tend to settle to earth around the explosion site and downwind from it soon after the explosion. Lighter particles may stay in the atmosphere for years. Radioactive decay products in fallout include strontium-90, potassium-40, carbon-14, and iodine-131. They may contaminate food supplies if taken up by plants and animals or contaminate water supplies by falling into streams. If they accumulate in the human body, they can form concentrated internal sources of dangerous radiation. Fallout may thus be a cause of leukemia, bone cancer, and other diseases. It can also cause genetic damage." I am stating that Morgellons Disease is the result of a combination of DMS, DMSP, marine particles and organisms, loosed genetically modified organisms, heavy metals; these micelles can incorporate a baculoviral component and other pathogens, etc., the vector is stratospheric and tropospheric fallout. That by the nature of DMS(P), it can easily be modified and pick up and carry anything it comes into contact with which can add to the Morgellons pathogen mixture. |
|
|
|
Post by kammy on Dec 8, 2010 15:54:50 GMT -5
culturingscience.wordpress.com/2010/07/19/dms/DMS(P): the amazing story of a pervasive indicator molecule in the marine food web "In order for water to transition from a gas to liquid in the atmosphere, it needs a small particle in the air to adhere onto, known as a cloud condensation nucleus. Sulfur oxide, which can be derived from DMS, is one of these particles. Clouds not only carry our precipitation, but help to reflect sunlight (and thus heat) back into space, affecting our planet’s climate. After the realization of its importance as a cloud condensation nucleus, scientists began to look for DMS’s planetary source and found that 95% of the atmospheric DMS originates in the oceans – but from where? The phytoplankton itself actually makes a molecule called DMSP (dimethylsuphoniopropionate, if you must know). When its cell wall begins to break down, stores of DMSP (from an unknown location within the cell) and an enzyme, DMSP-lyase, are released into the surrounding water. This DMSP-lyase removes the P-group, leaving us with our favorite molecule of the day, DMS. While the evidence is not directly causative, as the authors showed a reaction to acrylic acid and not to DMSP-produced acrylic acid in vivo, it does suggest (with other evidence) that the important part of this reaction to the phytoplankton is not DMS, but rather its byproduct. culturingscience.files.wordpress.com/2010/07/2010_07_18_dmsp_dms_vanalstyne2001.jpg?w=500&h=94But a paper published in Science this week (July 16 2010) by Justin Seymour, Rafel Simo, and others looks into the effects of DMSP on the smallest grazers: microbes. Using “microfluid technology” (see details at end of post), the researchers measured the strength of attraction of 4 different types of microbes (7 species) to varying concentrations of DMSP, DMS, DMSO (dimethylsulfoxide, a DMS and DMSP degradation product), GBT (glycine betaine, a molecule analogous to DMSP in structure and function), and artificial seawater (as a control). Each of the 3 species of autotrophic plankton reacted differently to DMSP in the water. The algae Micromonas pusilla showed strong attraction to DMSP, taking it up presumably as a carbon and sulfur source. The cyanobacterium Synechococcus sp. showed no reaction. Most strangely, the algae Dunaliella tertiolecta moved very strongly toward DMSP, but not to assimilate it directly; rather, it cleaved the molecule into DMS extracellularly and potentially assimilated that molecule instead. The authors do not know why this action occurs. Two species of bacteria, Pseudoalteromonas haloplanktis and Silicibacter sp., each moved toward the DMSP for assimilation as part of their carbon and sulfur requirements. Two consumers, one a herbivore to eat the algae itself (Oxyrrhis marina) and the other a bacteriovore which strove to eat the bacteria consuming the DMSP, each showed positive chemotaxis and moved toward the DMSP source. Thus far, we have DMSP attracting bacteria to consume the DMSP itself, drawing in an herbivore to consume the DMSP-producer, and a bacteriovore attracted to the DMSP in order to find its own bacterial prey. culturingscience.files.wordpress.com/2010/07/2010_07_18_dms_big_picture.jpg?w=500&h=646As all biogeochemists know, the stuff of the air frequently comes from the stuff we live on and in: soil and water. However, DMSP has something special: sulfur. We all know that smell, and perhaps it is this stinkiness that has allowed it to become such a pervasive indicator throughout the marine food web."
|
|
|
|
Post by kammy on Dec 8, 2010 16:06:11 GMT -5
Dimethyl sulphide biogeochemistry within a coccolithophore bloom tinyurl.com/2vlepjr"The process study comprised analyses of the time-varying biological, optical and physical properties of the patch as well as studies of DMS, dimethylsulphonioproprionate (DMSP), dimethylsulphoxide, nutrients, halocarbons, methylamines, carbon monoxide, dissolved organic carbon, and total dissolved nitrogen. The role of viruses, bacteria, phytoplankton, microzooplankton, and mesozooplankton, together with the dynamics of primary, new and bacterial production, plankton respiration, microzooplankton grazing, and sedimentation, were studied in relation to the biogeochemical cycling of DMS. Although the coccolithophore bloom water exhibited high optical backscatter, the algal community present was highly heterogeneous. Flagellates other than E. huxleyi were found to dominate the phytoplankton. A budget of the DMSP pools suggested that E. huxleyi accounted for only 13% of the stocks of particulate DMSP, showing that in this “E. huxleyi bloom”, taxa other than E. huxleyi were important sources of DMSP." |
|
|
|
Post by kammy on Dec 8, 2010 16:23:20 GMT -5
Coccolithophores: Earth's Little Helpers www.bbc.co.uk/dna/h2g2/A3709433"One of the more conspicuous effects is the long-term/short-term relationship of coccolithophores with carbon dioxide and global warming. Our problem: Humans annually release over six billion tons of carbon dioxide by burning fossil fuels and other activities, yet the scientists have found only three billion tons in the atmosphere; one can only wonder what has happened to the other half, for three billion tons of carbon dioxide cannot simply disappear without a trace. Solution: One coccolithophore is built up of numerous coccoliths, chalky, plate-like formations of calcite. Coccoliths are made up of one part carbon, one part calcium and three parts oxygen; CaCO3. Every time a molecule of coccolith is produced, one less carbon dioxide molecule is floating around as a greenhouse gas. Coccolithophores also consume carbon dioxide by photosynthesis. The majority of the 'vanished' carbon dioxide was sequestered by and converted into coccolithophores. Disguised Agents Can coccolithophores counter global warming? Maybe. Should scientists accelerate the growth of coccolithophores? These phytoplanktons have already removed tons of carbon dioxide by taking out the carbon to create the coccoliths. Supporters of coccolithophores' population increase should be aware that the same formation of coccoliths that has reduced the amount of carbon in the air also produces carbon dioxide molecules from oxygen and carbon in the oceans. Those molecules will most likely be used by coccolithophores, but some may escape back into the atmosphere. It is possible that, in the short-term, global warming could cause the upper layers of the ocean to become calmer and more stagnant, which would increase the population of coccolithophores, which in turn would increase the amount of greenhouse gas escaping into the atmosphere. Right now, however, the statistics show a consistent removal of carbon dioxide from the atmosphere (three billion tons a year). Coccolithophores also use up a vast quantity of bicarbonate ions in the ocean. Basically, the more coccolithophores there are, the more DMS gets released, and the more clouds are formed. The clouds mean more layers to block the Sun's rays, an increase in precipitation and overall, a cooler Earth. Nature's Thermostat Then there would be fewer coccolithophores left as there would not be enough energy for every coccolithophore to survive. Less DMS would be released, less clouds made for want of condensation nuclei, the sun would shine once more and lo, behold! In the oceans, the coccolithophores dwell once more in multitude... This DMS cloud formation cycle is repeated over and over again. This is how coccolithophores have helped Earth maintain stability at a generally tolerable temperature for millions of years. One coccolithophore species, Emiliania huxleyi or E. huxleyi, alone can cover more than 100,000 square kilometers3 of ocean surface and can produce tens of thousands of metric tons of CaCO3." |
|